Timik is the exclusive distributor of ZimVie Spine (formerly ZimmerBiomet) in the Nordic region. ZimVie is a global leader in spine innovation and develops top-class comprehensive thoracolumbar, cervical, biologics and implantable solutions intended to reduce pain, increase mobility, and restore function to daily life.
We deliver comprehensive thoracolumbar, cervical, biologics, and implantable solutions from ZimVie, along with training and clinical support.
The Mobi-C Cervical Disc has been designed for cervical disc replacement to restore segmental motion and disc height. The components of Mobi-C include superior and inferior cobalt chromium molybdenum alloy endplates coated with plasma sprayed titanium and hydroxyapatite coating, and a polyethylene mobile bearing insert.
ZimVie Spine utilizes its deep industry knowledge and expertise to refine a comprehensive anterior cervical product portfolio of next generation devices that includes Trabecular Metal™ Technology, PEEK and allograft options. The implants are designed to facilitate efficient, versatile, and reproducible spinal fusion procedures ready to meet the needs of surgeons and their patients in an ever-changing global spine market.

ZimVie Spine offers a full line of biologics products including advanced allografts, synthetic bone void fillers, and allograft demineralized bone matrix products to provide best-in-class biologic solutions for use in spine surgery.

ZimVie Spine’s comprehensive thoracolumbar products are engineered to treat spinal deformities, degenerative, and complex cases with clinical effectiveness, always with a commitment to patient safety.

Service is essential to keep medical equipment operational and safe. Our Nordic service team consists of experienced service technicians stationed across the Nordic countries. Our service technicians are ready to take on your every service need.”

Nasal PAP ventilation system
The SuperNO2VA nasal PAP ventilation system uses the flow from any oxygen source. Delivers positive airway pressure to stent open the upper airway, allowing for the preoperative delivery of positive pressure ventilation and oxygen for patients with a decreased level of consciousness. The SuperNO2VA™ nasal PAP ventilation device is available in medium and large sizes and is offered as a standalone mask with a head strap and as a system, kitted with a hyperinflation bag. These configurations offer flexibility as it allows the SuperNO2VA™ nasal PAP ventilation device to be used with either an anesthesia machine or with only an oxygen flow meter.

Sentec Digital Monitor (SDM) with activated PO2 channel for tcPCO2, tcPO2, SpO2, PR and HP monitoring
The Sentec Digital Monitoring System with PO2 (SDMS-PO2) provides continuous and non-invasive real-time monitoring of ventilation (tcPCO2), oxygenation (tcPO2) and Heating Power (HP) for neonatal patients. In pediatric / adult patients, the system can be used for tcPCO2, tcPO2, SpOI2, Pulse Rate, Pulsation Index (PI) and HP monitoring.
Transcutaneous monitoring (TCM) provides clinicians with greater insight to quickly identify trends, rapidly and accurately assess patient status, and make more well-informed, timely care decisions.
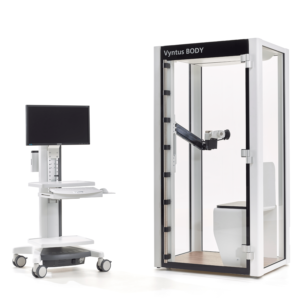
Vyntus™ BODY Plethysmograph
People come in all shapes and sizes, so we designed Vyntus BODY to accommodate the most patients without increasing the cabin footprint. Many enhancements were incorporated to make Vyntus BODY accessible and user friendly for both patients and technicians.
Vyntus BODY employs much of the same breakthrough technology found in the Vyntus ONE, but with a redesigned breathing circuit and our new Ultrasonic Sensor Technology.
In addition, Vyntus BODY is enhanced with additional innovation such as digital pressure compensation, which helps to reduce environmental influences and improves measurement accuracy.
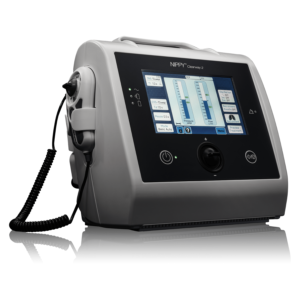
Versatile Airway Clearance
Introducing the Clearway 2 with TreatRepeat®. The second-generation MI-E device by Breas incorporates the latest technology to bring effective and accurate treatment for patients requiring airway clearance and cough assistance. TreatRepeat function allows clinicians to manually deliver different numbers of insufflations and exsufflations and then save the treatment they have just delivered to the device to be repeated automatically. Helping to improve accuracy and speed of titration and set-up.
Multiple mode options
Up to four treatment profiles can be saved to the device.The settings used in the manual and programmed modes are also saved to the device. This allows for up to 7 MI-E prescriptions to be saved. The treatment settings in the IPPB and NIV modes are also saved. The programmable mode enables the delivery of multiple insufflations as part of a cough cycle.