With POCcelerator® Data Management System, you can simplify your POCT workflows and dramatically reduce workload for your staff. Collect and review all patient results, quality controls, operators, reagents, and device data in a clear and structured format in order to comply with regulatory guidelines.
Maintain control of your POC Ecosystem™ Solution containing hundreds of devices, dozens of sites, and thousands of operators. Improve workflow, extend and enhance clinical capabilities, and improve the overall profitability of POC operations.
Open Connectivity
Connect over 220 POCT devices across 70+ manufacturers. Simplify your workflow and streamline your IT infrastructure by consolidating multiple middleware systems into one.
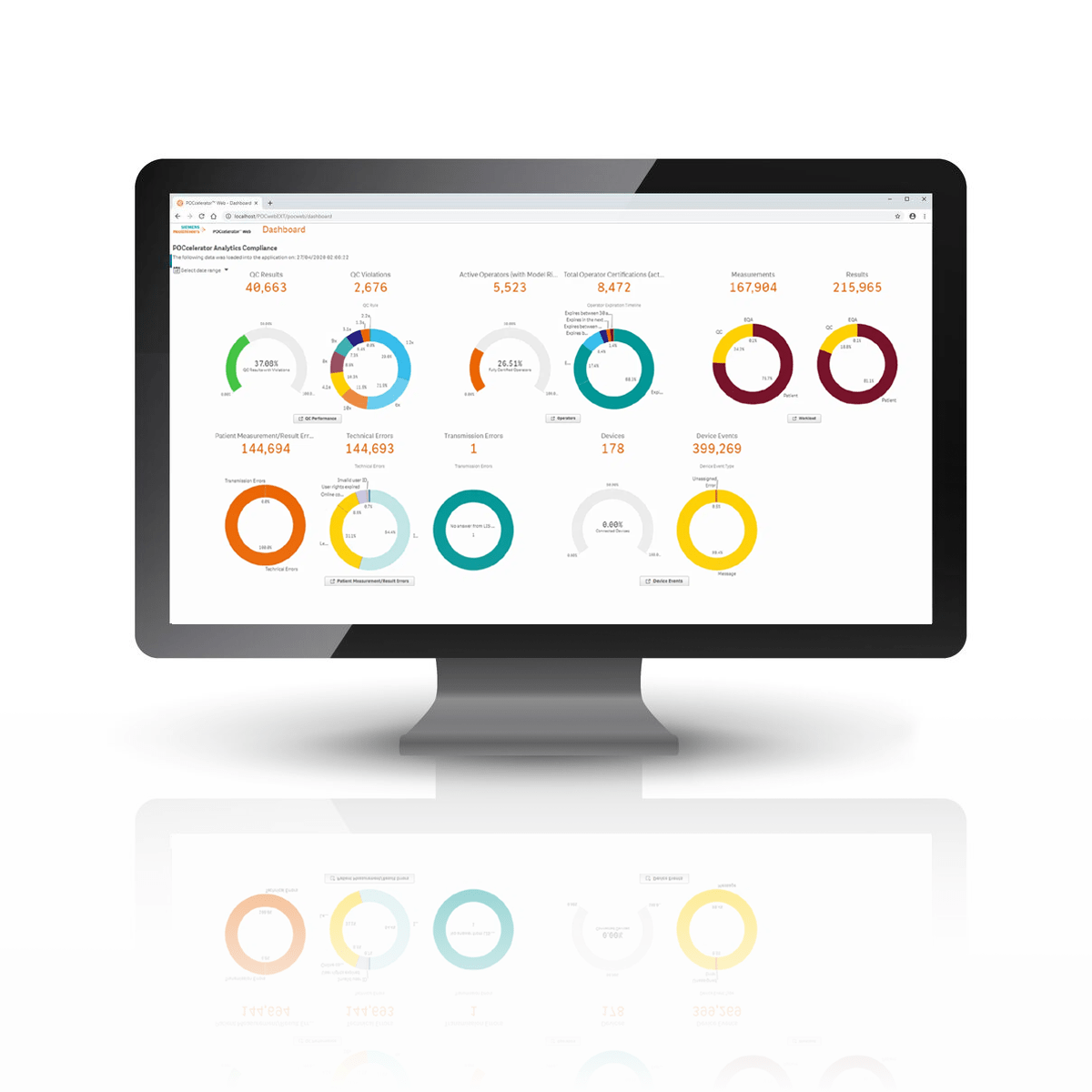
Recognize KPIs with POCcelerator Ci Module
At-a-glance metrics are available for Quality Control, Device Operators, Patient Results, and Device Performance. In-depth information is accessible with a few clicks.
Review trending reports
Analyze detailed reports
View your QC statistics for a visual representation of the selected criteria. Quickly assess any bias that may exist with different reagents.
Empower better care through deeper analytics and actionable insights
Every person, device, and POCT result must meet their respective standards of compliance. And you need to be able to monitor that compliance data and analyze it in real time to reduce risk and advance care. Data-driven, proactive, and efficient point-of-care testing is about more than quality control—it’s the gateway to better care.
Combining the most powerful release of the POCcelerator® Data Management System with in-depth, enhanced data analysis provided by the POCcelerator Ci module, delivers intelligent compliance to digitalize healthcare in ways previously unimaginable.
Interested in connecting only Siemens Healthineers devices? Ask your local sales representative about POCcelerator® SE Data Management System today!
Connect POCcelerator Data Management System to your Siemens Healthineers devices for maximum device performance
Download the ONE Solution infographic to see the enhanced features and functionality of your Siemens Healthineers devices when paired with POCcelerator System.

Atellica, Auto-Checks, CLINITEK Advantus, CLINITEK Status, DCA Vantage, epoc, Multistix, POCcelerator, RAPIDComm, RAPIDLab, RAPIDPoint, STIX, and all associated marks are trademarks – owned by or licensed to Siemens Healthcare Diagnostics Inc., or its affiliates. All other trademarks and brands are the property of their respective owners.
Service is essential to keep medical equipment operational and safe. Our Nordic service team consists of experienced service technicians stationed across the Nordic countries. Our service technicians are ready to take on your every service need.”

Meeting the needs of both medical staff and patient
The Vivo 65 is designed to meet the needs both of medical staff and a wide range of patients. It combines comfortable and controllable ventilation with excellent monitoring capabilities to potentially reduce readmissions to acute care facilities.
The Vivo 65 is an advanced homecare ventilator designed to deliver secure and comfortable (life-support) treatment to adult and pediatric patients from 5 kg. The Vivo 65 can be used for a wide variety of patients thanks to a comprehensive set of modes, circuits and accessories.
The dual limb circuit allows for measurement of exhaled volumes providing additional security. The extensive moni- toring capabilities help obtain a better insight into the quality of ventilation. The Vivo 65 is an excellent choice for mechanical ventilation at home, in the hospital and in long- term care facilities. The Vivo 65 is prepared for connection to Breas cloud solutions.

For accurate and safe bronchial challenge testing
Vyntus APS is an Aerosol Provocation System. Up-to-date and sophisticated electronics and mechanics allow for optimal use of its nebulization technology with precise dosing. For the observation measurements Vyntus APS combines with the Vyntus PNEUMO and/ or the Vyntus IOS in one system.
APS nebulizer system integrates into Vyaire MasterScreen™ and Vyntus™ instruments – Spirometry, IOS Impulse Oscillometry, Rocc and BODY Plethysmography. The nebulizer system is powered by SentrySuite™ software and is able to test a broad age range: from 4 years to adult. Choose between single and multiple concentration testing.

Helping heal one step at a time
From rehabilitation to orthopedics to physical therapy, the PPS (Professional Power System) Med treadmill provides comfort and assistance to patients who require walking support. This treadmill can accommodate any user’s pace with an absolute zero start speed with increments of 0.1 mph.
Vyntus™ BODY Plethysmograph
People come in all shapes and sizes, so we designed Vyntus BODY to accommodate the most patients without increasing the cabin footprint. Many enhancements were incorporated to make Vyntus BODY accessible and user friendly for both patients and technicians.
Vyntus BODY employs much of the same breakthrough technology found in the Vyntus ONE, but with a redesigned breathing circuit and our new Ultrasonic Sensor Technology.
In addition, Vyntus BODY is enhanced with additional innovation such as digital pressure compensation, which helps to reduce environmental influences and improves measurement accuracy.