PARI SINUS2 offers a unique form of nebuliser therapy for the treatment of acute and chronic sinusitis. PARI SINUS2 causes the aerosol to pulsate so that it can penetrate your sinus cavities. Right there where it is needed.
Aerosol characteristics
MMAD: 3.0 µm
Mass percentage below 5 µm: 66.6 %
Minimum filling: 2 ml
Maximum filling: 8 ml
Aerosol characteristics of PARI LC SPRINT SINUS nebuliser was measured with the PARI SINUS2 compressor. Particle size was measured with the Next Generation Impactor (NGI). Measurement at 23 °C and 50 % relative humidity. Nebulised medium 2 ml Salbutamol (1 mg /1 ml) Jet Flow 4.6 l /min.
Compressor
Electrical connection: 220V – 240V / 50 Hz
Case dimensions (W x H x D): 18.5 x 13.0 x 15.0 cm
Weight: 1,7 kg
Spare parts: All spare parts can be purchased from your local PARI retailer.
The PARI SINUS2 also turns saline solution or the prescribed medications into an aerosol. But only the PARI SINUS2 generates a pulsating aerosol. Thanks to the pulsation, it can go all the way into your nasal sinuses. In this video, you will see how to use the PARI SINUS2 correctly.
Service is essential to keep medical equipment operational and safe. Our Nordic service team consists of experienced service technicians stationed across the Nordic countries. Our service technicians are ready to take on your every service need.”
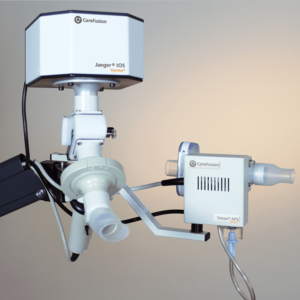
IOS Impulse Oscillometry
Tidal breathing analysis with Impulse Oscillometry (IOS) has demonstrated to be informative and differentiated in the early detection and follow up of pulmonary diseases like asthma, COPD and idiopathic pulmonary fibrosis. IOS is almost independent of patient cooperation and can test a larger patient range than spirometry alone, from children to adult to geriatric patients.
IOS is available as a stand-alone device combined with a spirometry measurement program (MasterScreen IOS and Vyntus IOS) or as an add-on module to the MasterScreen series.

The dual-valved LiteAire MDI holding chamber: Collapsible, disposable paperboard design.
The LiteAire® collapsible MDI holding chamber is a unique MDI holding chamber and an innovative alternative. LiteAire’s unique dual-valved MDI holding chamber design delivers pop-up convenience and effective drug output at a fraction of the cost of most plastic holding chambers. In many clinical settings, the LiteAire can reduce costs by replacing existing rigid plastic holding chambers or inefficient spacers with a paperboard alternative. The unique design allows the LiteAire to be reused by a patient over multiple doses and meets and often exceeds the performance of plastic holding chambers.
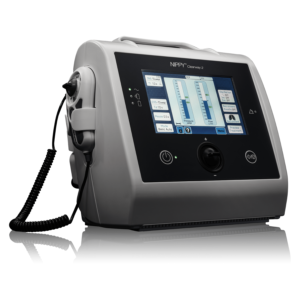
Versatile Airway Clearance
Introducing the Clearway 2 with TreatRepeat®. The second-generation MI-E device by Breas incorporates the latest technology to bring effective and accurate treatment for patients requiring airway clearance and cough assistance. TreatRepeat function allows clinicians to manually deliver different numbers of insufflations and exsufflations and then save the treatment they have just delivered to the device to be repeated automatically. Helping to improve accuracy and speed of titration and set-up.
Multiple mode options
Up to four treatment profiles can be saved to the device.The settings used in the manual and programmed modes are also saved to the device. This allows for up to 7 MI-E prescriptions to be saved. The treatment settings in the IPPB and NIV modes are also saved. The programmable mode enables the delivery of multiple insufflations as part of a cough cycle.
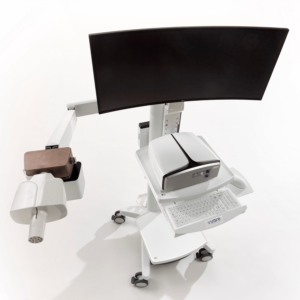
The power of ONE – Vyntus ONE is our innovative pulmonary function system equipped with an ultrasonic sensor.
Vyntus ONE is incredibly capable with a modular design that possesses an impressive array of features. Your Vyntus ONE can even grow later on together with your needs delivering high performance and utility in a single package. It is even able to be extended to also measure cardiopulmonary exercise tests with the same module.
It is designed to make your tests more accurate, less intrusive and less stressful for patients.